Introduction
In 2022, a survey was conducted in Italy with the goal of evaluating the effects and safety of a specific specialized pro-resolving mediators (SPM) supplement on people with chronic pain. The survey involved 70 nutritionists who each worked with one to five subjects over a period of three months, during which the subjects took the supplement daily and completed a questionnaire about their symptoms at the start, one month in, and three months in.
The SPM supplement was in the form of softgels. Subjects took 2 softgels a day for the first month and then 1 softgel a day for the second and third months. The questionnaire asked the subjects to rate the severity of their pain on a scale of 0 to 10 (0 = no pain; 10 = extreme pain) and how much the chronic pain impacted various aspects of their lives on a scale of 0 to 10 as well (0 = no impact; 10 = extreme impact). Of the 70 nutritionists involved, 48 finished the survey with at least one subject, and 39 of the 48 finished the survey with at least two subjects. In total, there were 189 subjects (71% women and 29% men) who participated, with ages ranging from 17 to 87 years old and an average age of 53. The most common areas of pain reported were the core body (40.7%), followed by the legs (34.9%), arms (14.3%), and head (10.1%). Out of all the subjects, 149 completed the first two months and 120 completed the full three months of evaluation.
Survey results
Pain level over the last two weeks
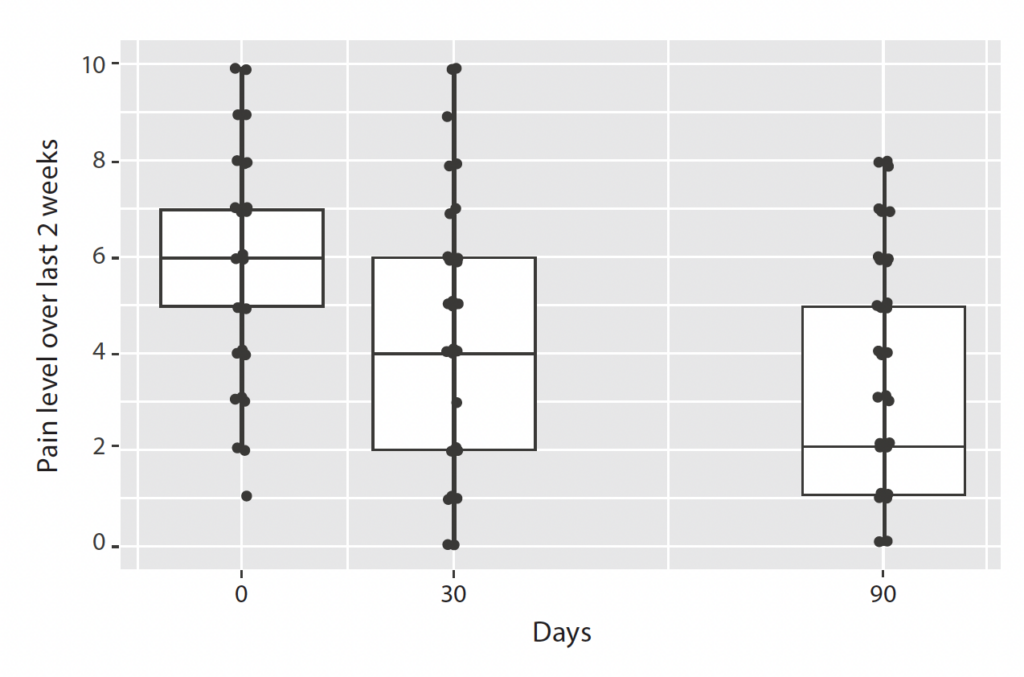
At baseline, one month, and three months, subjects gave a score of their average pain level during the last two weeks. The distribution of the scores is depicted in Figure 1. The wide distribution of pain scores (black dots) at baseline indicates subjects experienced a wide range of pain levels when they joined the survey. Data from one and three months indicate that there is a clear trend toward reductions in pain levels as a group (but not everyone): The median pain score decreased from 6 at baseline to 4 after 30 days and to 2 after 90 days, and scores for the interquartile range (the range for the middle 50% of the group, shown in boxes in Figure 1) also improved over time.
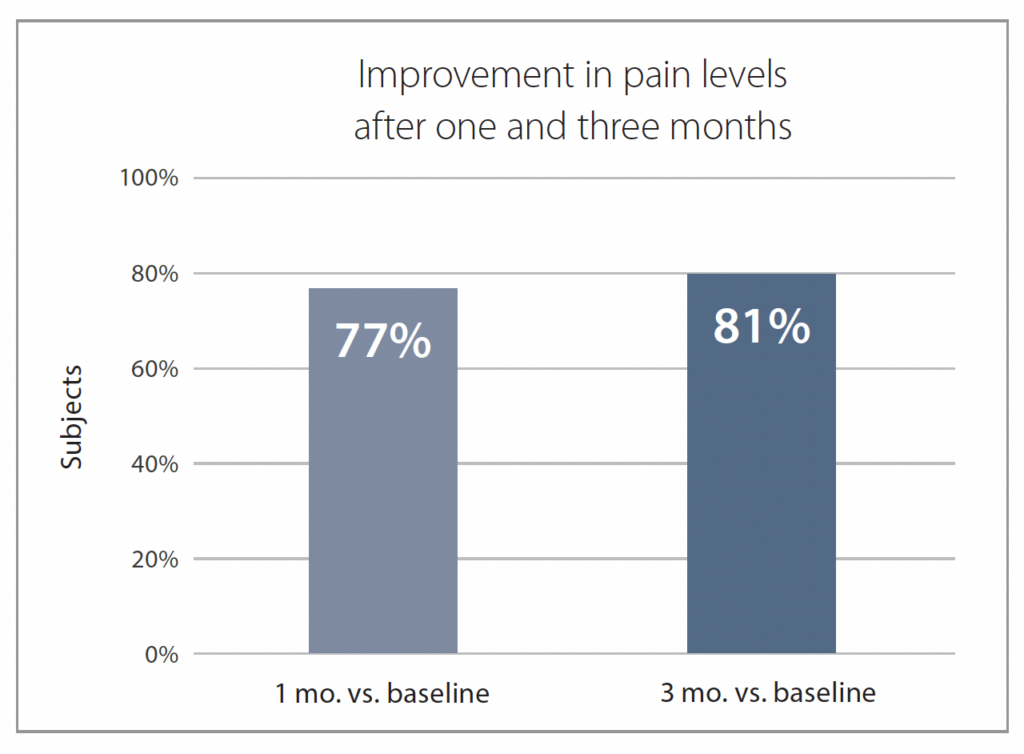
After one month, 77% of participants reported less pain compared to baseline, and this number increased to 81% after three months (Figure 2).
Quality of life
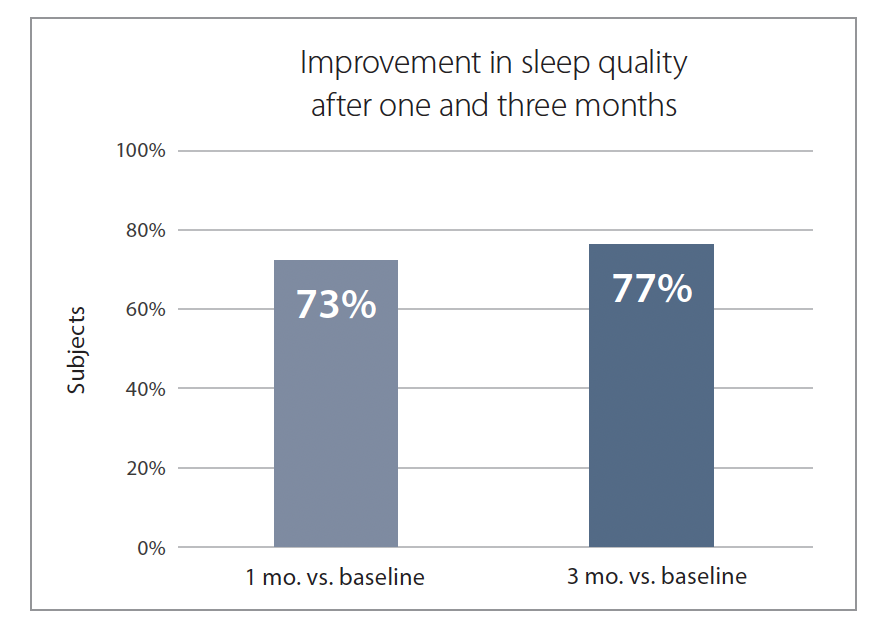
The questionnaire asks how, during the past 24 hours, chronic pain impacted their quality-of-life measures such as sleep, mood, and normal work. After one month, 73% of participants reported better sleep quality, and this number increased to 77% after three months (Figure 3).
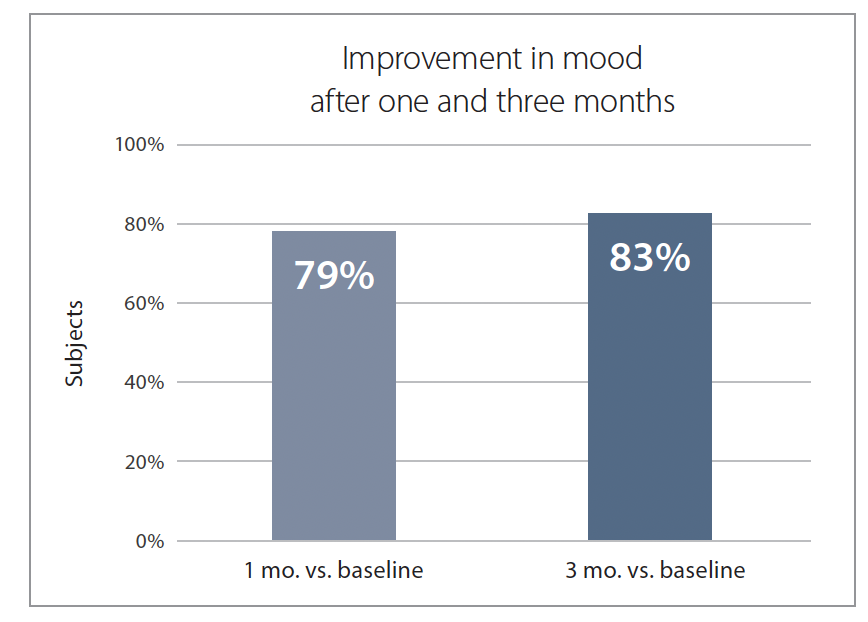
Nearly 8 out of 10 participants (79%) reported an improved mood after one month, and this increased to 83% after three months (Figure 4).
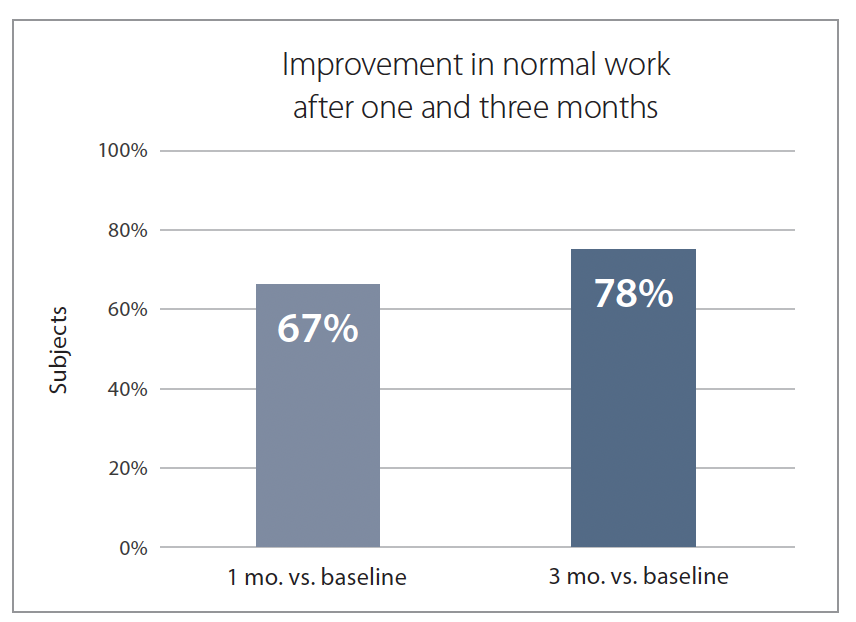
More than two out of three participants (67%) experienced an improvement in their normal work after one month, and this number increased to 78% after three months (Figure 5).
Product satisfaction
Regarding the ease of swallowing the SPM supplement, 96% of those surveyed answered positively. Around 1% reported experiencing minor side effects, such as slight heartburn. In terms of the clarity of the product description and its use, 98% responded positively. Finally, when asked if they would continue to use or reuse the product, 93% of the participants indicated they would.
Summary
In this postmarketing survey, conducted in Italy in 2022, the effects and safety of a SPM supplement on people with chronic pain were evaluated. Subjects took the supplement daily and completed a questionnaire about their symptoms at the start, one month in, and three months in. Results showed a clear trend toward pain reduction and improvements in quality-of-life measures such as sleep, mood, and normal work. Additionally, most participants reported satisfaction with the product and indicated they would continue to use it.


